



Every day the majority of people around the world will come into contact with an object that has been improved through electroplating. Unfortunately, even with this level of adoption, outside of the industry, very few understand what electroplating is, what the process entails, and how it improves surface materials. Before deciding whether electroplating is right for your product, it’s best that you understand a few things about the electroplating process, what to consider about the part being plated, the material options available, and your goals.
Electroplating, or electrolytic plating, is a technique in which a thin layer of metal is deposited, using an electric current, onto an object to enhance the surface characteristics. The actual specifics on electroplating are a bit technical. A process called hydrolysis distributes atoms evenly, merging the two materials. There are a number of different types of metals that can be used, including copper, silver, titanium, nickel, and chromium.
Just as there are many variations in electroplating, electroplated goods have different uses, too. Sometimes the coated surface is simply decorative, as in a collectible; other times it’s functional, as when we work with industries that use industrial machinery, processing equipment, and vehicles used in all modes of transportation. Sometimes the surface of a part or object needs to be hard to prevent wear, other times it needs to be porous; sometimes it needs to be matte with low reflectivity, other times shiny.
The first electroplating process was invented over 200 years ago by Italian chemist Luigi Valentino Brugnatelli. It wasn’t until the late 1830s that electroplating would be developed into a major industrial process, and as electroplating and the science behind it advanced, new commercial uses were developed using metals like nickel, tin, zinc, and brass.
Towards the late 19th century, a breakthrough technology changed the electroplating landscape. Electric generators, able to carry higher currents, allowed for production level processing of parts. This new level of throughput ability highlighted the benefits of electroplated components in automotive, machining, and other major industries. Hastened development, during the two world wars and the birth of the aviation industry, saw increasingly new processes such as hard chromium electroplating, sulfamate nickel plating, and many others. The number of chromium electroplating facilities around the United States grew substantially during this time, including what would become the first U.S. Chrome in 1940.
Today, there is continuous improvement and innovation in the electroplating industry as new metal finishes and processes emerge. The global push to reduce the use of certain chemical processes, which are deemed environmentally unsafe, has started a surge for alternative coatings.
Electroplated goods have many different uses. The process can be used to create a decorative finish that gives a high-value feel and look. It can also be used to create a finish that’s functional and has uses in machinery and equipment. Listed below are the uses for electroplating.
Aesthetics
The most common use of electroplating is for aesthetic purposes, where a thin layer of expensive metal is applied to a product made from an inexpensive metal. This gives the product the desired look while keeping the cost low. An example of using electroplating for aesthetic purposes would be jewelry. Pieces of jewelry can be made from common metals at a reasonable price. They can then be electroplated with gold, silver, or other rare metals which will add the desired look to the final product.
Corrosion Protection
Depending on the environment, all metals will corrode. Metals naturally react with the chemicals in the environment and over time these reactions can cause corrosion like rust. Adding a layer of non-corrosive plating finishes, like tricom, nickel, or chromium, will protect the substrate from corroding. These electroplated coatings will break down less quickly and increase the life of the product.
Surface Hardness
Mainly seen in industrial applications, electroplating can be used to create extremely hard surfaces. Metals that are under the stress of heavy use need to be able to withstand that stress. If a soft metal is used without this added hardness, it could flex and possibly break. Chromium electroplating will harden the surface between 65-70 HRc.
Reduced Friction & Wear
When two metal objects are designed to come into contact with each other there is going to be friction between them. Over time, this friction at the contact points will cause wear. There are a number of metals that can be electroplated onto the surface of these objects to reduce the friction, increasing the life and performance.
Electrical Conductivity
Electrical components, used in electronic goods such as cell phones and computers, need to use metals that are good conductors of electricity, like gold and silver. Having components made out of those two metals would be very expensive though. Electroplating allows for the components to be made out of less expensive metals and then coated with a thin layer of gold or silver to achieve the needed conductivity.
Buildup Thickness
Engineers that are designing parts to work within tight tolerances will purposefully have parts machined undersize and then sent for electroplating. Electroplating allows for precision buildup, controlling the final size.
Electroplating is a material additive process in that it deposits material to the surface of an object or base metal. Before further detailing the process, the four essential parts of an electroplating system need to be explained.
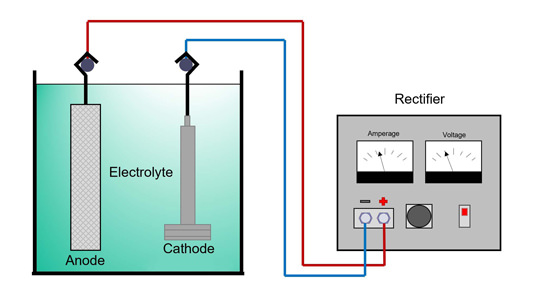
Shown in the image above are the rectifier, electrolyte, anode, and cathode.
Rectifier
A rectifier is a device that receives power from the electrical lines we see outside and converts the incoming alternating current (AC) to a direct current (DC) that will be used in the process. An important part in the electroplating process, the rectifier transforms the high voltage, low amperage AC supply, that is not suitable for most metal finishing operations, to a substantially reduced voltage and increased amperage DC supply.
Electrolyte
The electrolyte is an aqueous ionic solution comprised of many ingredients that assist in the electrodeposition process. Some of these ingredients include the metal or metals that are being deposited and conducting salts that promote good electrochemical stability.
Anode
In most cases, the anode, or positively charged electrode, is made of the same metal being deposited onto the object. In other cases, like with chromium, the anode will only be used to transmit the electrical current. In these cases, the anode needs to be made out of a material that will not dissolve or deteriorate, like lead.
Cathode
The cathode is the object to be plated. It is a negatively charged object that attracts the ions reduced at the anode.
Now that we know these four essential parts, we can explain the process, which starts with the preparation of the cathode (object). The areas on the object that require plating need to be prepared in a way that allows for the proper adhesion of the material that is being added. This includes masking off areas that cannot be plated, chemical pre-treatments, and rinses to wash off any remaining chemicals, dirt, or debris that could affect the plated surface.
The liquid bath is then prepared containing various ingredients required for successful plating of the desired material. The temperature and chemical composition of the electrolyte bath needs to be monitored throughout the process. The object and the anode are placed into the electrolyte bath and connected to the rectifier. It’s important to note that the placement of the anode, and the object, in the bath, as well as the geometries of the object, can result in thicker or thinner deposit areas.
Once the electrolyte bath is electrified, the metal at the anode will oxidize, turning atoms into positive ions. Those newly formed positive ions will migrate to and bond with the object because it is negatively charged. Over a certain period of time, this will create a thin layer of perfectly adhered metal.
To explain this process in practical terms, we can look at electroplating hard chrome onto a hydraulic rod. The electrolyte would contain chromic acid, the anode would be made of lead, as it only used to direct the electrical current, and the hydraulic rod would be the cathode (object). Both the lead anode and the hydraulic rod would be placed into the electrolyte and power from the rectifier would be supplied through the lead anode. The lead anode would reduce the chromic acid in the electrolyte to positively charged ions, that would then bond to the surface of the hydraulic, creating a layer of hard chrome.
Thickness of the deposit can be manipulated via changes to the amperage being supplied at the rectifier and the length of time the work piece stays in the bath. The coating is deposited on the surface areas of the workpiece that are directly exposed to the positively charged inert anode. This method can build heavy deposits for repair and salvage work.
While there are a variety of different electroplating methods, all of which will achieve similar results, two are more commonly used. Those methods are rack plating and barrel plating. Both of these options have advantages and disadvantages, but the use of each is dependent on the application to be plated.
Rack Plating
Racking plating is used for parts that are large and/or have geometries that are difficult to plate. Parts are attached to a metal rack and connected directly to the rectifier. The rack is placed into the electrolyte bath where the plating process will occur. While rack plating is more expensive, this method will apply a higher quality coating. This process would typically be used for aerospace components, medical instruments, and oil and gas parts.
Barrel Plating
This method is used to plate a high volume of small parts at a time. Parts are placed into a large barrel that will be filled with one of many different types of electrolyte solutions. During this process, the barrel is rotated, moving the parts so that they receive an even finish. Barrel plating is typically more cost effective than rack plating. While barrel plating has a very high throughput, it is not recommended for parts that require a precision, engineered finish. With so many parts being plated at once, there is a high chance that the finish could have minor imperfections that would affect the usability.
Many government agencies and industries use electroplating and have specific composition requirements. For example, a locomotive crankshaft requires different elements than, say, landing gear on a fighter jet. Specific parameters change from country to country. If you’re working in an industry with an institutional body or governmental regulations, it’s best to know what that body needs before making your electroplate selection. To see some of the industries we work with and parts that we electroplate, visit our industries page.
Process
Remember that the thicker the chrome plating, the more difficult to maintain the object’s tolerance and surface finish. See the table below for guidelines. Also, remember to define the surface finish.
Application
Where exactly will the chrome plating be applied? There are many techniques that can be employed to direct the plating to the needed surface. Electroplating requires electrical contact be made with the object. Contact points receive little or no plating coverage.
Usage
Will the object requiring plating encounter abrasive surfaces or elements, or will it be exposed to high heat or corrosive environments? Do you need conventional coating or are there special needs, like crack-free or oil retentive?
It’s also important to keep in mind that electroplating does not hide defects. In actuality, it’s the opposite: Any dents, dings, pits, scratches, or other surface anomalies will be exaggerated, so be sure to examine the surfaces carefully prior to submitting for electroplating.
Suggested Thickness and Tolerance Hold for Electroplating
| Condition | Thickness | Tolerance |
| Light, corrosive atmosphere | 0.0002-0.0005 thick | 0.001 |
| Most wear application | 0.0007-0.0015 thick | 0.001 |
| Water and chemical attack | 0.002-0.005 thick | 0.002 |
We’re confident US Chrome will be your best option for your chrome plating applications because of our engineering approach, exceptional customer service, and regional locations. Contact us now with your questions or coating challenges.
We’ll help you find the right answer.